Yanislava Karusheva, Matt Ratcliff, Audrey Melvin, Alexander Moerseburg, Naveed Sattar, Peter Barker, Keith Burling, Anna Backmark, Robert Roth, Lutz Jermutus, Esther Guiu-Jurado, Matthias Blueher, Paul Welsh, Marko Hyvönen, Stephen O’Rahilly
The Journal of Applied Laboratory Medicine, Advance Article 07 July 2022.
DOI: 10.1093/jalm/jfac055
MedRxiv preprint DOI: 022.01.03.22268655
Abstract
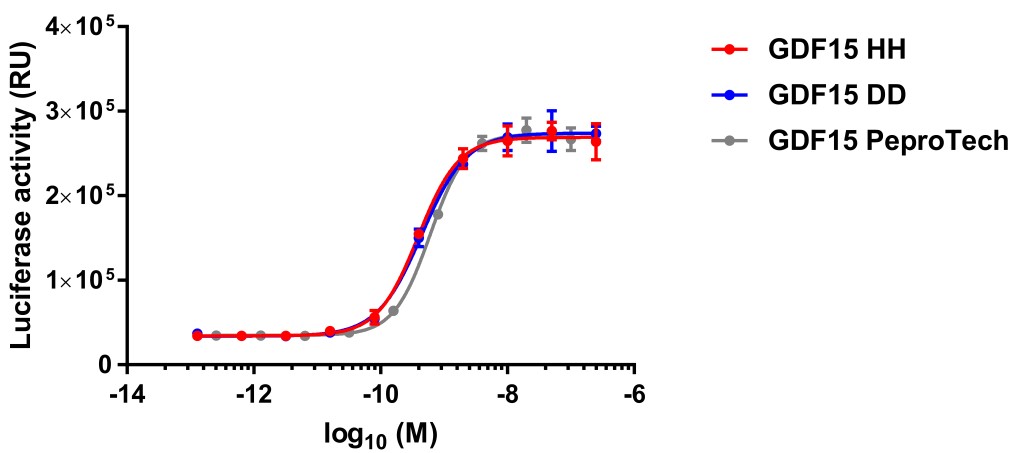
Genetic variants in proteins can interfere with measurement of their circulating concentrations. Given the growing biomedical importance of GDF-15, we wished to establish whether a common histidine to aspartate variant present in position 6 of the mature GDF-15 protein (H202D variant) interfered with its measurement by two commonly used immunoassays. We first examined the detectability of recombinant monomers, homodimers and heterodimers of GDF-15 by assays and/or reagents used in two widely used immunoassays (Roche Elecsys GDF-15 and the R&D antibody combinations used in their Quantikine and DuoSet ELISAs). The Roche assay detected the H and D containing peptides similarly but the assays based on the R&D reagents consistently underreported concentrations of the D-containing variant peptide. Measurements of plasma concentrations of GDF-15 in genotyped human participants showed that the R&D reagents reported values in heterozygotes were ~25% lower, and in homozygotes, 50% lower than the Roche assay. We finally studied the activation of the GDF-15 receptor, GFRAL-Ret, in a cell based assay and found that the activities of the HH and DD containing GDF-15 peptide were indistinguishable. These results have implications for the interpretation of genetic epidemiological studies which have used the R&D reagents to measure GDF-15, and for the emerging clinical use of GDF-15 as a diagnostic and prognostic biomarker. We provide correction equations, which may be of utility for the analysis of data generated with the R&D reagents where the genotype of the participants is known.
